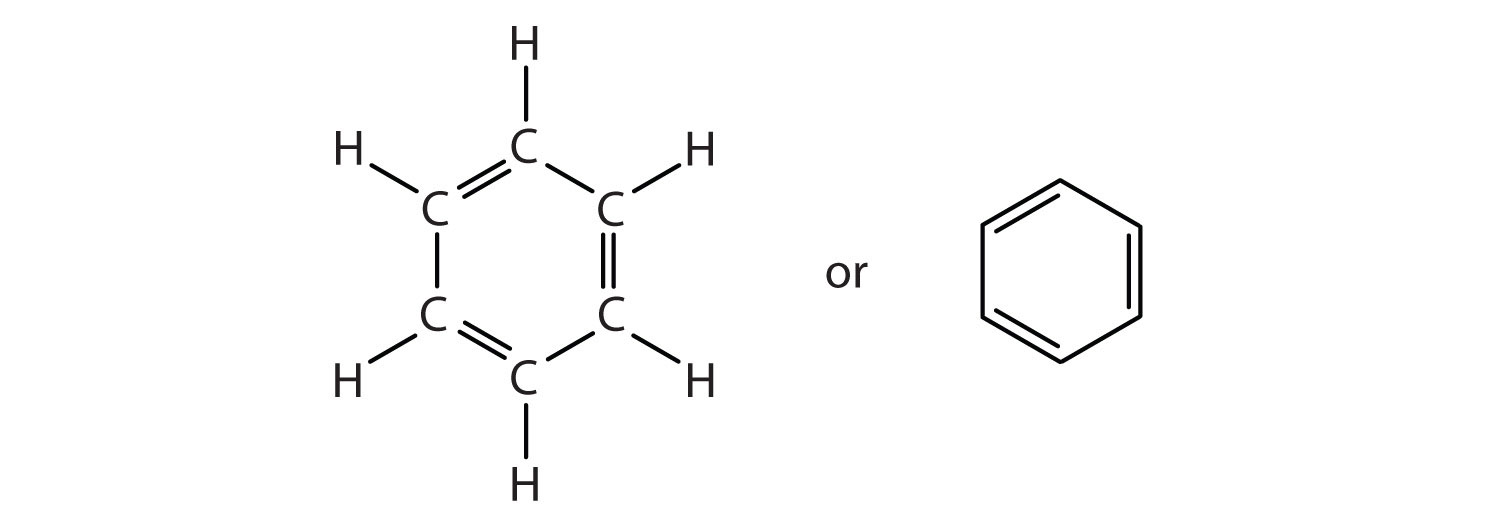
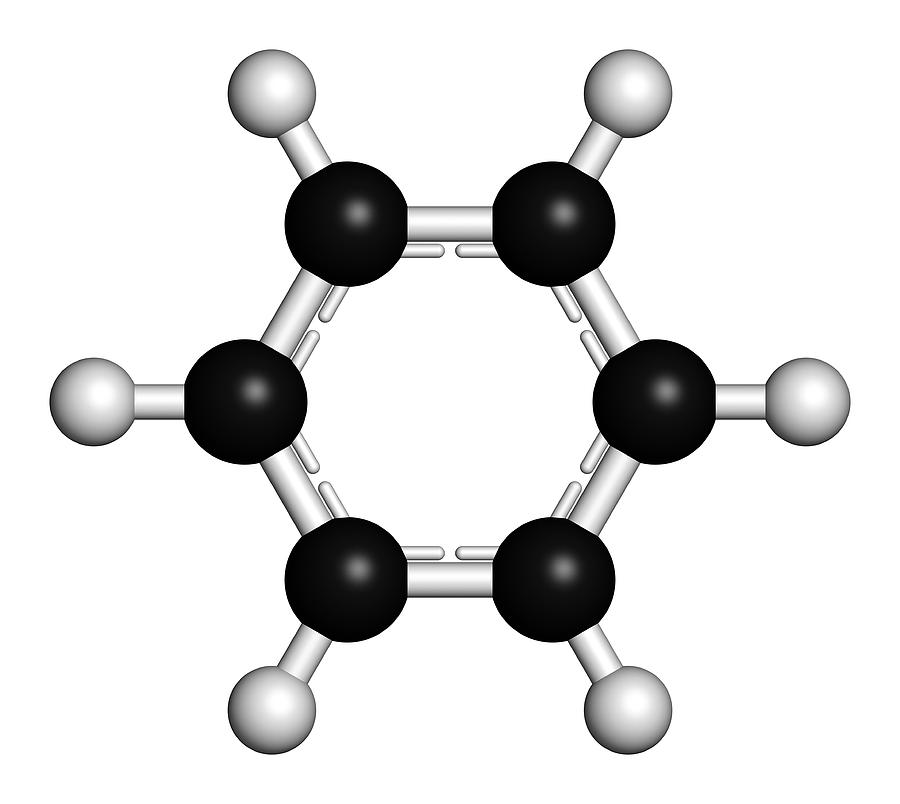
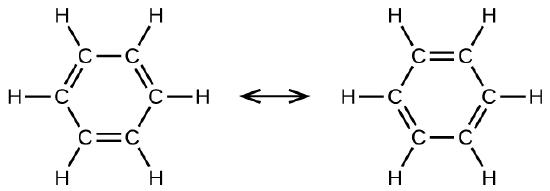
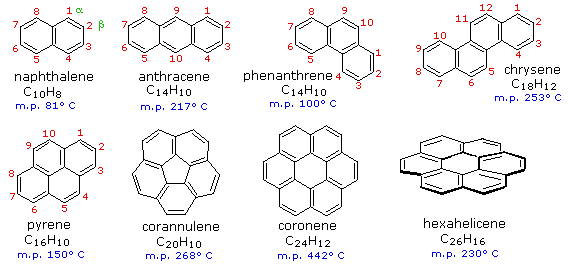
.png--diagram-flowchart-example.png)
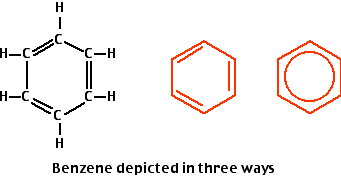
Design elements - Aromatic hydrocarbons (arenes) | Phenols | Aromatics - Vector stencils library | Aromatic Hydrocarbon Reaction
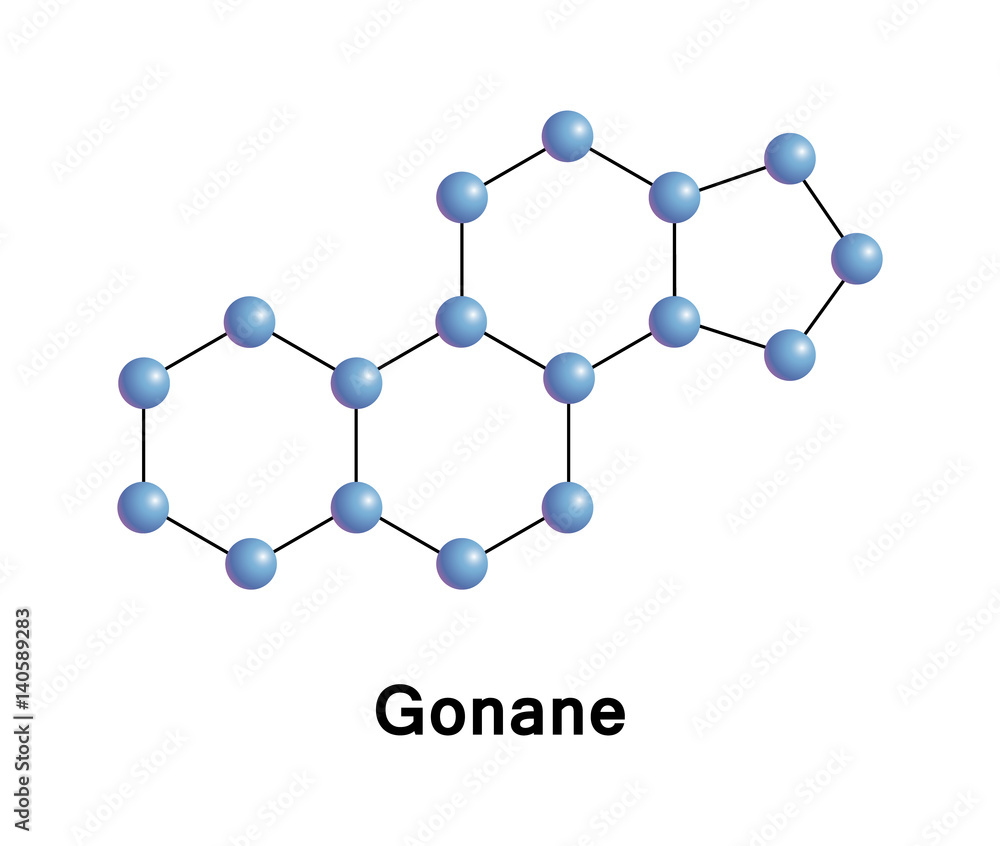
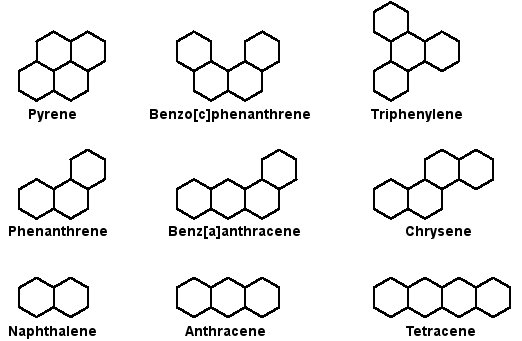
Gonane, known as perhydrocyclopentaphenanthrene, is a tetracyclic hydrocarbon ring structure and the fundamental steroid nucleus. It consists of a phenanthrene ring fused with a cyclopentane ring Stock Vector | Adobe Stock

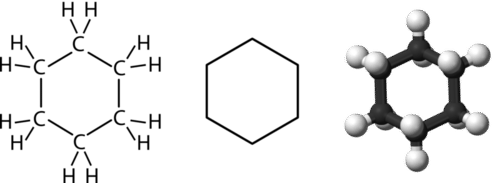
Benzene Chemistry Aromatic hydrocarbon Friedel-Crafts-Acylation Deuterium, benzene ring, sphere, chemistry png | PNGEgg

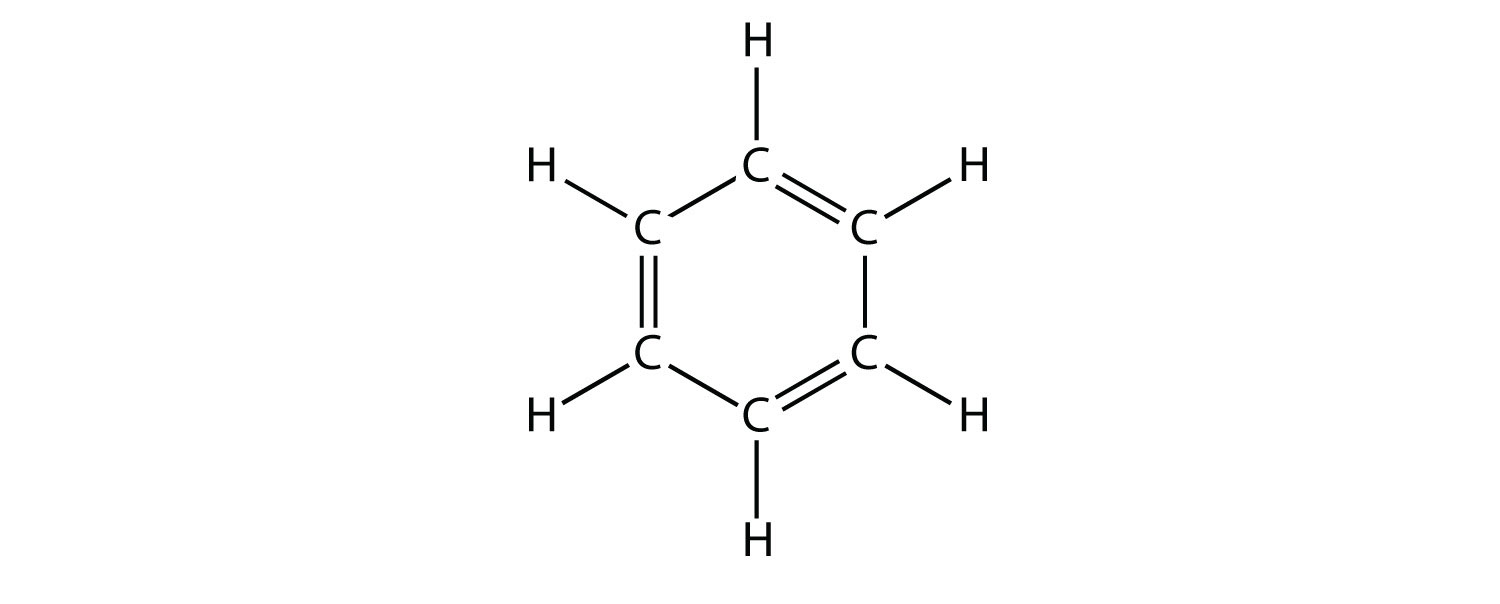
A hydrocarbon consists of a benzene ring and two methyl groups. The methyl groups are located at opposite carbons and the molecule is perfectly symmetrical. Which option correctly describes the numbers of